Examining the basic principles of quality by design (QbD) approach in analytical studies. - Document - Gale Academic OneFile

Development and Optimization of Liquid Chromatography Analytical Methods by Using AQbD Principles: Overview and Recent Advances | Organic Process Research & Development
QbD Approach Method Development for Estimation of Dabigatran Etexilate along with Its Impurities and Identification of Degradants in Capsule Dosage Form

Development and Optimization of Liquid Chromatography Analytical Methods by Using AQbD Principles: Overview and Recent Advances - ScienceDirect
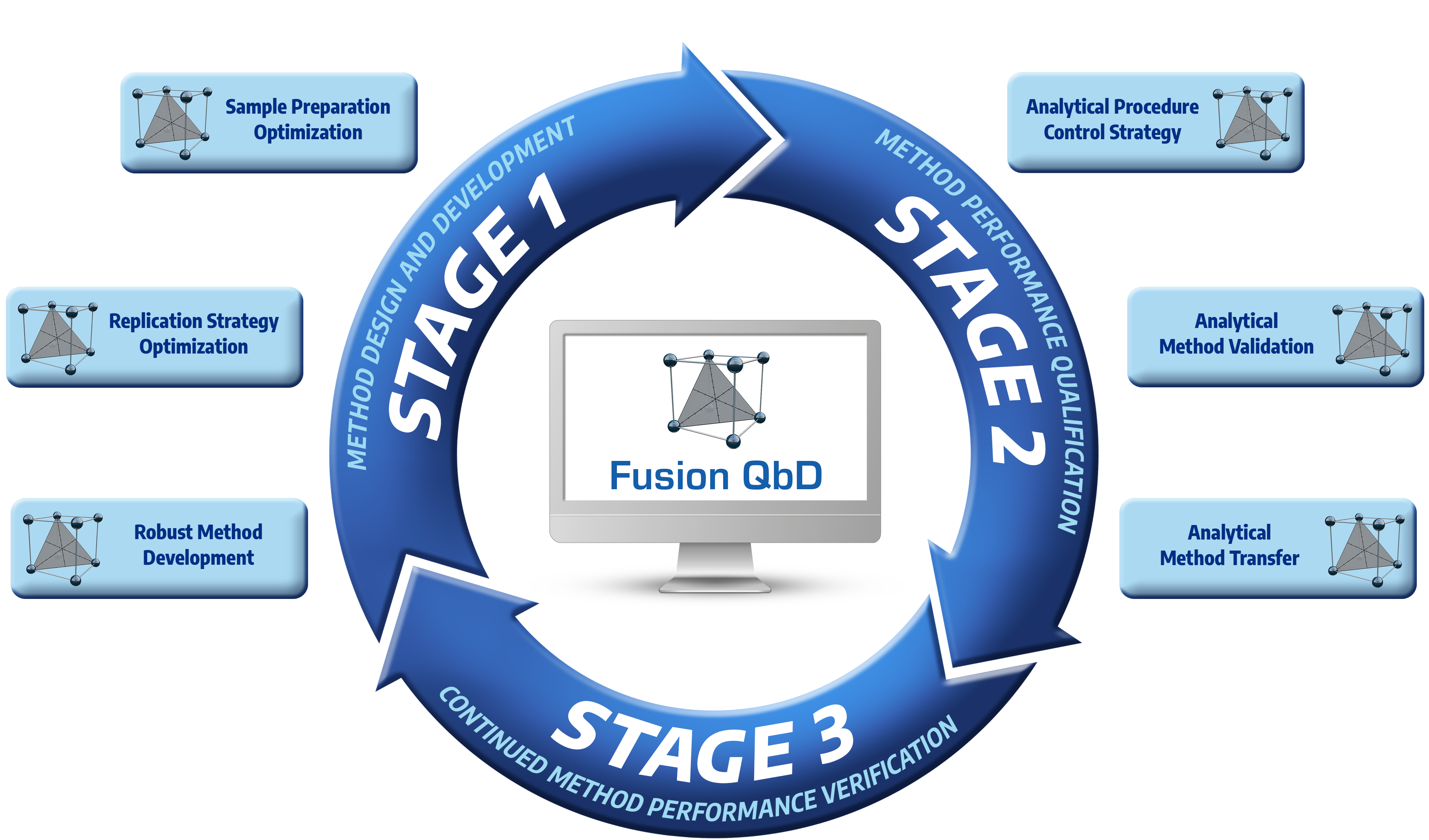
Fusion QbD Software - Quality by Design - DOE - Automated Data Analysis - Chromatography Application Software

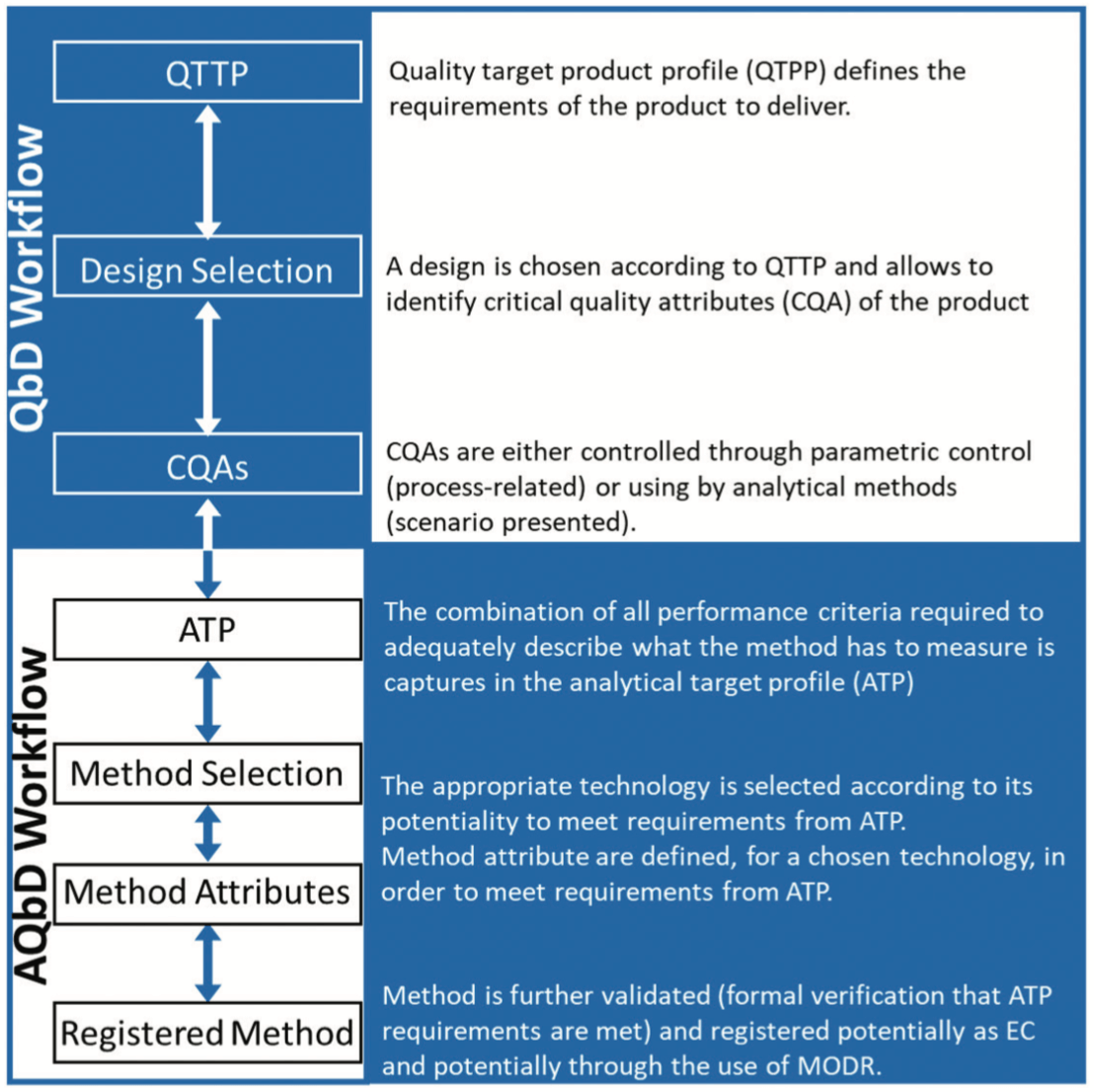
Analytical Quality by Design Approach to Test Method Development and Validation in Drug Substance Manufacturing

An analytical quality by design (aQbD) approach for a l-asparaginase activity method - ScienceDirect

Development and Optimization of Liquid Chromatography Analytical Methods by Using AQbD Principles: Overview and Recent Advances | Organic Process Research & Development

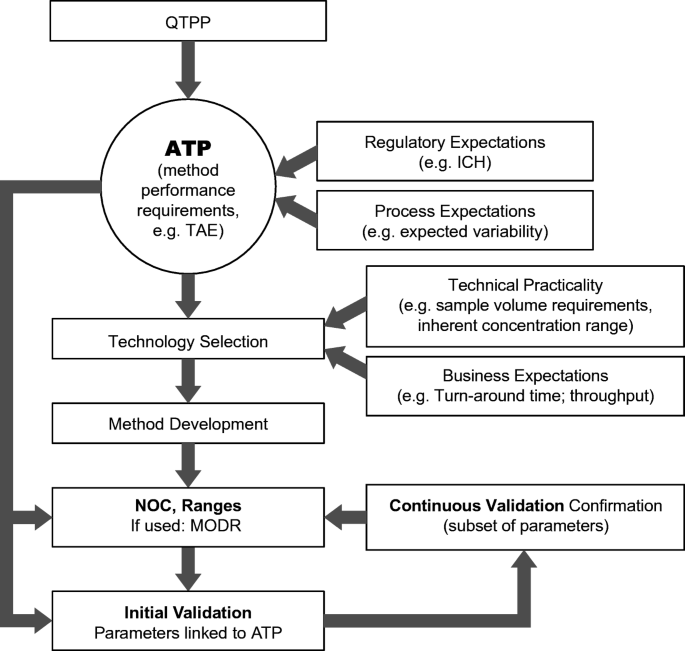
Figure 1 from Analytical Quality by Design Approach to Test Method Development and Validation in Drug Substance Manufacturing | Semantic Scholar
Analytical method development by using QbD - An emerging approach for robust analytical method development

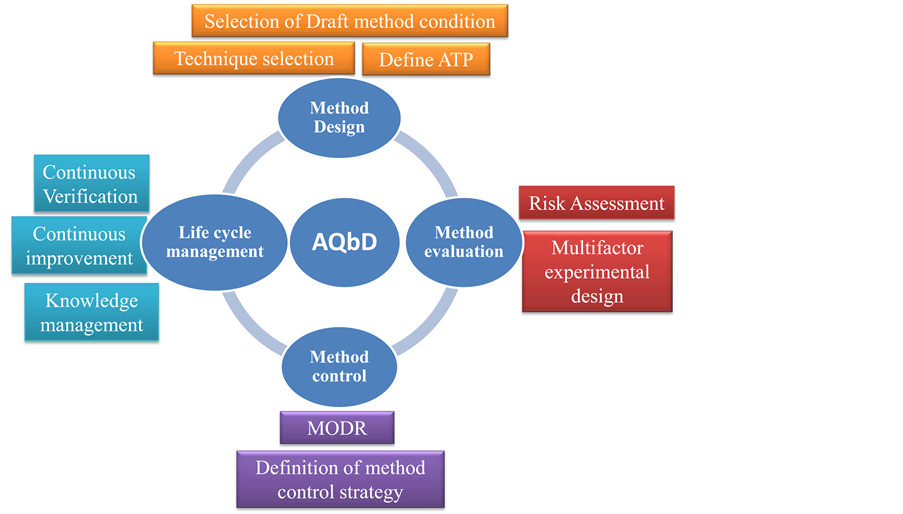
Figure 2 from Quality Improvement with Scientific Approaches (QbD, AQbD and PAT) in Generic Drug Substance Development: Review | Semantic Scholar

Maximize lab productivity and quality by embracing expected ICH Q14 Enhanced Method Development Guidelines and AQbD approaches Registration